Bailey Williams suffered hundreds of seizures a day before cannabis oil gave him his life back – but his family is forced to raise thousands to fund it. Bailey’s mum, Rachel Rankmore tells Sarah Sinclair why they won’t stop fighting for NHS access to the drug.
When the coronavirus lockdown came into force earlier this year, Rachel Rankmore and her husband Craig Williams’ first thought was whether they would still be able to import the cannabis oil that gives their son a better quality of life.
The second was how they would continue to raise the £1,700 each month to fund the prescription.
Bailey Williams, 19, has a rare form of epilepsy, Lennox Gestaut Syndrome.
He was born a healthy baby, but at the age of two and a half his parents woke in the night to the sound of him choking – Bailey was experiencing his first fit.
Medics initially diagnosed a febrile convulsion, but over time the seizures became more frequent – and severe – and Bailey was put on anti-epileptic drugs.
“Your whole world comes crashing down,” says Rachel, 44, a full-time carer for Bailey at their home in Cardiff.
 “We were first-time parents and you imagine that you’re going to have this perfect family.
“We were first-time parents and you imagine that you’re going to have this perfect family.
“Bailey had to stop going to nursery, the seizures became more violent and progressed into drop seizures, absence seizures, myoclonic jerks, and partial seizures happening throughout the day and night.
“He had to wear a crash helmet and afterwards would be left bedridden or in need of a wheelchair.”
Bailey tried over 20 antiepileptic drugs, all of which failed to stop the seizures and left him experiencing extreme side effects, including hair and weight-loss and head-to-toe rashes.
“He had a really traumatic time on the medicines that were supposed to make him well and they just weren’t working,” continues Rachel.
“The quality of life was just awful for the whole family.”
Then the couple’s second son Ross, born four years later, was also diagnosed with a less severe form of epilepsy as a toddler.
Rachel and Craig made the decision to stop his medication, which was making him fall asleep at school, and thankfully Ross, now 14, has been seizure-free since.
But both boys have been left with learning difficulties and dyslexia as a result of their condition.
The family went onto try and control Bailey’s epilepsy through a ketogenic diet, which left him at risk of diabetes, and even explored brain surgery which he wasn’t a candidate for.
After what Rachel describes as ‘years of hell’, Bailey was given a vagus nerve stimulation, aged seven, which is supposed to prevent seizures by sending regular, mild pulses of electrical energy to the brain via the vagus nerve.
Although it alleviated some of his symptoms, Bailey being blue-lighted to hospital in an ambulance was still a weekly occurrence.
At their lowest point, with Bailey enduring another stint in the High Dependency Unit at the University Hospital of Wales, doctors told Rachel and Craig they had nothing left to try.
“The doctor said we have given him everything we can…we don’t know if he’s going to wake up at all and we’ve got nothing left to try,” she says.
“We just felt total despair.”
But they kept looking for other options, and came across the story of Charlotte Figi – the American youngster who helped popularise CBD use for the treatment of her epilepsy.
 Charlotte’s Web CBD, named after the girl was founded in 2011. And when a friend of Rachel’s discovered a place in the UK was selling it, around six years ago they didn’t hesitate.
Charlotte’s Web CBD, named after the girl was founded in 2011. And when a friend of Rachel’s discovered a place in the UK was selling it, around six years ago they didn’t hesitate.
Rachel says: “We got in the car and went straight to buy our first bottle of Charlotte’s Web. That was a major turning point for Bailey, it gave him his life back.”
The drops seizures stopped, he became more vocal and was able to start learning again at a special needs school.
She adds: “He was just like an average boy, it was absolutely amazing.”
But just over two years ago Bailey started to plateau on Charlotte’s Web – which is not a full extract cannabis oil – and began deteriorating rapidly.
At one point he was having hundreds of seizures a day.
Around the same time, Hannah Deacon was fighting for access to cannabis for her son Alfie.
After seeing her story on This Morning, Rachel and Craig joined other parents in the End our Pain mission to change UK law. Alfie became the first patient in the UK to receive a permanent cannabis licence and is now only one of less than patients with an NHS prescription.
But despite being a candidate for medical cannabis Bailey’s doctors wouldn’t prescribe it.
Guidelines published by the National Institute for Health and Care Excellence (NICE) last year recommend Epidyolex – a drug containing CBD with clobazam – to be offered as an option used to treat seizures associated with Lennox-Gastaut syndrome in children aged two years and older.
Rachel says they were offered Epidyolex but had heard from other parents it had failed to be effective and instead turned to a private neurologist in London, who prescribed Bailey full extract cannabis oil.
The starting dose cost £2,500 and to reduce the cost of thousands more in fees incurred by importing the drug from Holland, Rachel criminalised herself, travelling to the country in person to collect Bailey’s medicine.
They now rely on donations through the Mission for Bailey’s Shakes fundraising page – initially set up to raise £55,000 to buy an EEG machine for the hospital – supporters have continued to raise money to cover the costs of Bailey’s prescription, becoming “close friends” along the way.
“Bailey’s quality of life is amazing, he’s able to interact and tell us what he wants to do, he wants his own home and he wants to drive and he’s coming up with all these things that he wants to do in the future, which is incredible and quite emotional because it’s something we never thought we’d see,” says Rachel.
But Rachel and Craig, who still take turns to sleep next to Bailey every night in case he has a seizure, fear what the future looks like for their son if cannabis oil is not made available on the NHS.
“It’s a treatment, it’s not a miracle cure but it’s given Bailey his quality of life back and we never want to take that away from him, that would be cruel,” she continues.
“But we can’t sustain the level of costs indefinitely – what happens when we’re not here?”
The coronavirus lockdown has meant they have been unable to hold fundraising events, such as fairs, golf tournaments and bike rides which keeps the Mission for Bailey’s Shakes fund topped up and have now switched to a cheaper UK supplier.
The struggles of the previous months have made the family more determined in the fight to see cannabis oil available to patients on the NHS.
“It would be amazing for us to have an NHS prescription so we can just live our lives like everyone else, without having to think where the next lot of money was coming from,” says Rachel.
“It would be such a weight off our shoulders to know that we can plan a future for Bailey, to be able to stop fighting and fundraising and just enjoy our son being well.”
She adds: “This is why we need to keep fighting to make sure our children are safe and well on the medicine that works for them.”
NICE has acknowledged the need for more research into the use of medical cannabis and supports NHS England’s call to collect evidence from randomised controlled trials and observational studies.
But the guidelines state that until there is clear evidence of the safety and effectiveness of cannabis-based medicinal products, specialist doctors need to consider individual patient circumstances and risks and benefits in choosing treatments.
As most cannabis-based medicinal products are currently unlicensed, doctors must follow General Medical Council guidelines of prescribing.
A spokesperson for Cardiff and Vale University Health Board, said: “Our clinicians always act in the best interests of patients and put them at the centre of everything we do. We will continue to work with Bailey and his family to best manage his condition.”
Donate here https://uk.gofundme.com/f/mission-for-baileys-shakes

 News6 months ago
News6 months ago
 Science5 months ago
Science5 months ago
 Industry6 months ago
Industry6 months ago
 News6 months ago
News6 months ago
 News5 months ago
News5 months ago
 Health5 months ago
Health5 months ago
 News5 months ago
News5 months ago
 Health3 months ago
Health3 months ago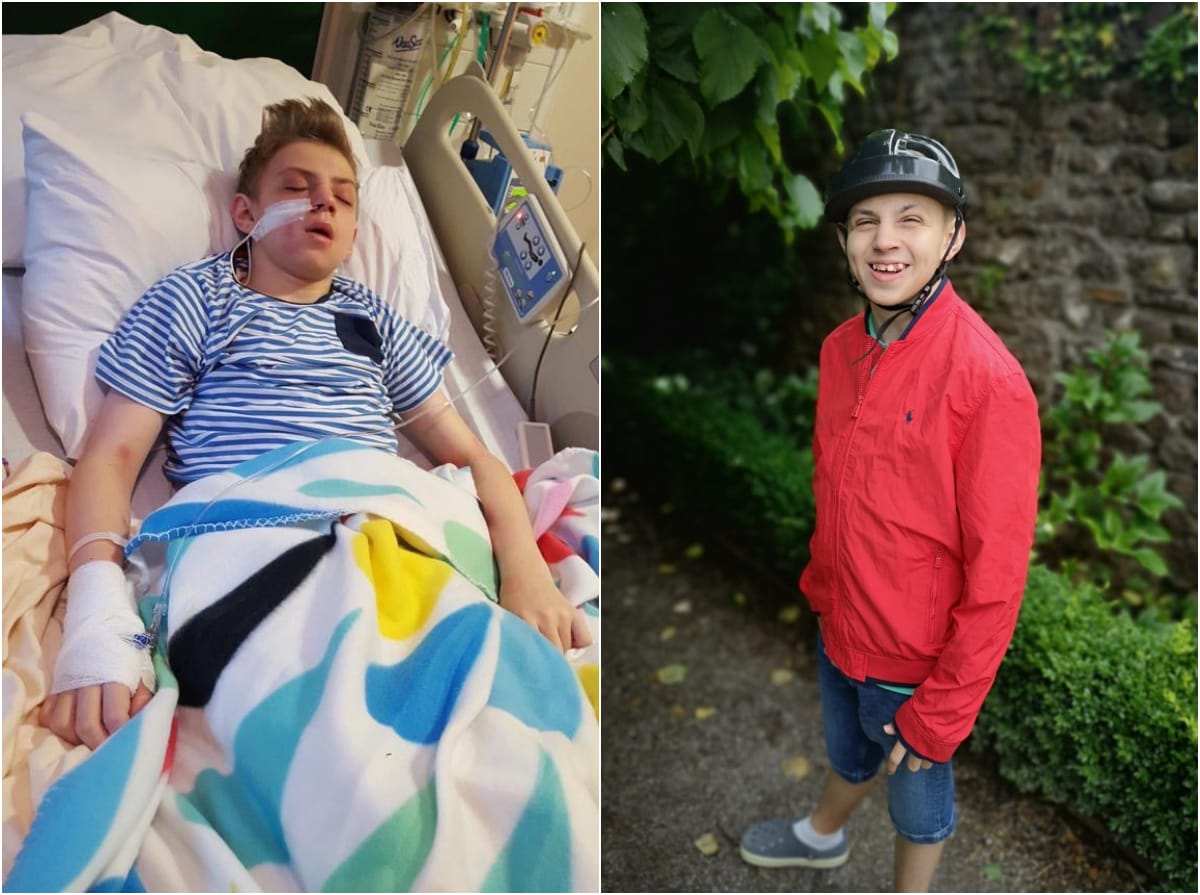

 Charlotte’s Web CBD, named after the girl was founded in 2011. And when a friend of Rachel’s discovered a place in the UK was selling it, around six years ago they didn’t hesitate.
Charlotte’s Web CBD, named after the girl was founded in 2011. And when a friend of Rachel’s discovered a place in the UK was selling it, around six years ago they didn’t hesitate.











